-
基金项目
- 国家自然科学基金资助项目(2137616.
作者简介
-
文章历史
-
2015-11-25 收稿
2016-01-04 录用
2016-01-14 网络版发表.
实践证明水中的疏水性物体之间存在引力,这种引力称为疏水引力.它在诸多反应中扮演着重要作用,例如胶束形成和生物薄膜聚合等[1],为了从源头弄清楚疏水引力的起因,研究人员用原子力显微镜或表面力仪测得两个疏水物体间受力和距离的关系曲线,并根据这些力-距离曲线提出了许多关于疏水力来源的理论,在这些理论中最可信的就是纳米气泡桥理论,这个理论基于凝聚态物理界的一个新发现,即溶解在水中的气体会在疏水性表面吸附和积聚[2, 3, 4, 5, 6, 7, 8]. 由此,学界认为疏水物质之间的引力是由吸附在疏水性表面的气体相互联通而形成的纳米气泡桥引起的[9, 10, 11, 12, 13, 14, 15, 16]. 这一观点已经被广泛认可,但是关于纳米气泡桥的形成过程和形态还没有一个清晰的描述. 本文的任务就是采用分子动力学方法来计算石墨烯间的疏水引力并通过分析气体分子分布规律的变化来证实纳米气泡桥是疏水引力的根源,并详细表述其形成和消失过程.
1 模 拟本文采用的是分子动力学模拟方法,其优势在于可以准确控制各种实验条件并且可以记录所有分子的运动轨迹、密度分布和总能量的变化情况.
1.1 理论依据用分子动力学研究物体间的相互作用,最直观的方法就是看它们之间的势能随距离的变化,因为势能对距离的导数就是力(F=dE/dS),在分子动力学中这叫做平均力势(PMF,potential of mean force)法,它的原理基于概率分布. 假设系统有A和B两个状态,通过统计系统在状态A上的时间tA和在状态B上的时间tB,求系统在状态A上的概率PA =tA/(tA+tB)和系统在状 态B上的概率PB=tB/(tA+tB),得到平衡常数K=PB/PA,AB两个状态 的势能差就可以通过Delta Gº-RTln K求得.然而,真实系统不可能只有A和B两个状态,而是无数个状态.处理方法是沿着作用方向把坐标切成若干小段,将其离散化.让系统内的分子自由移动,最后统计系统落在每一段上的时间比例,这样得到的是一个概率密度分布,然后换成每一小段(状态)上的势能变化,连成一条起伏的势能变化曲线,即平均力势曲线. 因为横坐标是作用距离,纵坐标是势能变化,因此曲线各点的斜率就是作用力.但是,由于分子动力学模拟本身的缺陷. 模拟过程中系统一般不会自动访问所有的状态,特别是高势垒的状态,这样会造成结果采样不足,最终无法统计. 只能采用伞形抽样方法,具体做法是在作用物体上施加偏势并进行偏采样,再通过加权直方图分析法(weighted histogram analysis method,WHAM)把偏采样的统计结果转换为无偏采样的统计结果,得到平均力势曲线.
1.2 初始模型因为要模拟一对石墨烯片在有气体溶解和无气体溶解的水中的相互作用. 模拟的初始模型如图1所示,5nm× 5nm×20×nm的模拟盒子按照每立方纳米填充33个简单点电荷(simple point charge,SPC)水分子模型作为水 (每立方纳米33个水分子符合300,K温度时水的宏观性质),碳原子数为240的两片正方形石墨烯片被平行地放置在水中 间,间距为0.5nm. 有气体溶解的情况,采取在水中均匀的混合2000个氮气分子.

|
图1 分子动力学模拟的初始模型 Figure.1 View of the simulation system |
原子间的受力采用如下方法计算:形成共价键的原子,其受力情况由谐振势V(r)=Kb(r-b0)2/2描述,其中Kb和b0分别代表键常数和键的标准长度.对于没有成键的原子间的受力由兰纳-琼斯(Lennard-Jones)势函数[17]U = 4εij [( σij/r)12-(σij/r)6]描述,其中ij和ij是原子之间的距离和能量常数,这两个参数遵从如下计算法则:σij=(σii+σjj)/2,εij=(εiiεjj)1/2.气体和固体的力场参数如表1所示,水的力场遵从安博96 (Amber 96) [18]力场.
| 表1 固体和气体的力场参数 Table 1 Parameters of force-field of solid and gas |
模拟的第1个阶段是能量最小化. 之后,系统需要经过两个阶段的平衡.即第2个阶段的恒体积(NVT)平衡,在这阶段水和气体分子用贝伦德森(Berendsen)弱耦合法 [19]使温度保持在300,K;和第3个阶段的恒压(NPT)平衡,在这个阶段同样用布德森弱耦合法[19]使压力保持在1.0×105,Pa. 在这前3个阶段内,两片石墨烯片都被固定在原来的位置上不动.接下来的第4个阶段是分子动力学模拟阶段,A石墨烯被固定,B石墨烯处于自由状态,并且用欧兹-胡 沃(Nose-Hoover)恒温器 [20, 21]维持温度不变,用帕内罗-拉赫曼(Parrinello-Rahman)恒压器[22, 23]维持压力不变,这种组合可以真正保证模拟为恒压(NPT)模拟.第5个阶段是加载偏势拉动B石墨烯的阶段,在这个阶段中,把A石墨烯固定不动,用一个正弦势把B石墨烯拉开到距离A石墨烯较大的位置. 第6个阶段是选出各段伞状抽样 [24, 25]的初始构象,首先从第5个阶段模拟的轨迹文件中提取出对应不同石墨烯间距的中间构象,再按照一定的采样距离从这些构象中找出若干个构象作为伞状抽样 [24, 25]分段采样的初始构象.第7个阶段是让这些初始构象平衡,这个平衡阶段两片石墨烯都被固定,其参数设置与第4个阶段类似.第8个阶段是对各初始构象进行模拟并分段采样,这个阶段中A石墨烯被固定,B处于自由状态. 第9个阶段是用加权直方图分析法获得平均力势曲线 [26].
2 分析与讨论 2.1 没有气体溶解的情况经过前4个阶段的模拟,我们发现两片石墨烯的间距从初始的0.5,nm缩小至约0.37,nm.在这个过程中石墨烯不受任何外力,间距变化是自发完成的,这说明石墨烯之间的相互吸引是自发完成的.
第5阶段(拉动阶段)用正弦势把两个石墨烯拉开,拉开速度为2pm/ps,正弦势的力常数为10MJ/(mol-nm2), 两片石墨烯的距离从0.37,nm拉开到1.37,nm,并从中提取了500个中间构象,从这500个构象中按照0.1,nm的采样距离选取出11个构象,作为下一步伞状抽样过程中的各采样区间的初始构象.其中,间距为0.5,nm的初始构象和间距分别为0.5,nm,0.55,nm,0.67,nm,0.8,nm,0.9,nm,1.0,nm和1.1,nm的中间构象以及这些构象中水的密度分布被表示在图2中.从中可以很清楚地看出:当石墨烯间距小于约0.5,nm时(如图2(b)所示),两片石墨烯之间自发形成了一个空穴(其中没有水分子),类似结论在文献[27, 28, 29]中也有介绍;当石墨烯间距大于约0.5,nm时,水分子开始进入空穴,形成一个水蒸汽的纳米气泡桥(如图2(c)图2(f)). 当石墨烯间距大于约0.8,nm时,空穴中的水分子(水蒸气)吸附在石墨烯表面形成水膜.关于这个水膜,相关研究证实它是由于受到固体表面分离压力的影响而形成的,其性质呈现出受限液体的特性,即类固体的特性[30];同时,也有研究证实水在两个疏水表面之间确实会有一些相变发生[31].由此可以看出在水膜之间就又形成了一个二阶水蒸汽纳米气泡桥(如图2(g)图2(h)). 进而,当石墨烯间距大于约1.1,nm时,水蒸汽纳米气泡桥消失了(如图2(i)).
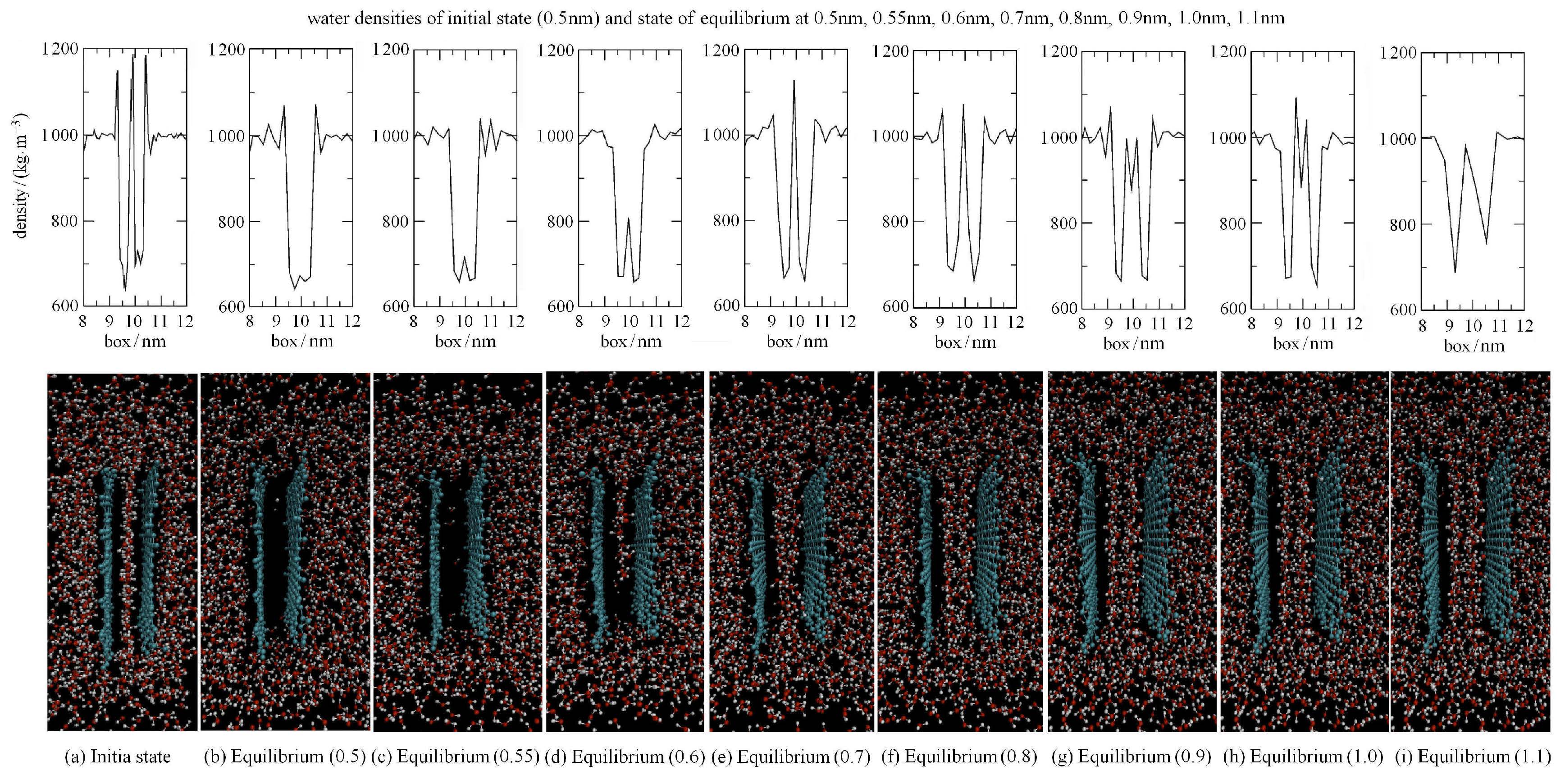
|
图2 初始构象和部分中间构象图及其对应的水密度分布 Figure.2 Water densities and corresponding configurations at different |
在伞状抽样后,通过加权直方图分析法得到了平均力势曲线(如图3). 由于曲线各点的斜率变化表征疏水力变化,从图3可以看出疏水力随石墨烯间距增大而减小;并且,在0.37∼0.8nm之间的斜率明显大于0.8∼1.1nm之间的斜率. 这表明0∼0.8nm间的疏水力是由一阶水蒸汽纳米气泡桥引起的,0.8∼1.1nm间的疏水力是由二阶纳米气泡桥引起的. 当石墨烯间距大于约1.1nm时平均力势曲线水平,说明气泡桥断裂,疏水力消失.

|
图3 没有气体溶解的情况,两片石墨烯在水中相互作用的平均力势曲线 Figure.3 The calculated PMF curve for two pieces of graphene in the expansion without gas phase |
经过前4个阶段(时长11ns)的模拟,同样发现了两片石墨烯的间距从初始的0.5nm缩小至约0.37nm.另外,溶解在水中的气体吸附在了两片石墨烯上(如图4所示).

|
图4 不同距离的构象和对应的气体密度分布 Figure.4 Gas densities and corresponding configurations at different distances |
拉动阶段(第5阶段)所用时长为2ns,拉开速度为2pm/ps,正弦势的力常数为5000kJ/(mol-nm2),两片石墨烯的距离从0.37nm拉开到4.37nm,并从中提取了800个中间构象,再从这800个构象中按照采样距离约为0.05nm选取出81个作为伞状抽样的初始构象. 其中的部分构象图和对应的气体密度分布如图4所示.从图中可以很清楚地看出,当两片石墨烯的间距小于约0.5nm时,两片石墨烯之间同样自发形成了一个空穴,其中既没有水分子也没有气体分子 (如图4(a) );当两片石墨烯的间距大于约0.5nm时,气体分子开始进入空穴,形成一个低密度纳米气泡桥;直到约0.8nm,空穴被气体充满形成了一个饱和气体纳米气泡桥(如图4(b) );之后,随着两片石墨烯的间距的增加,水分子开始挤压气体分子(如图4(c)∼ 图4(f) )气泡桥呈现中间细两端粗的形态;直到约3.8nm时,纳米气泡桥从中间断裂(如图4(g) ).由此,可以推测纳米气泡桥的形成和消失过程如图5所示.
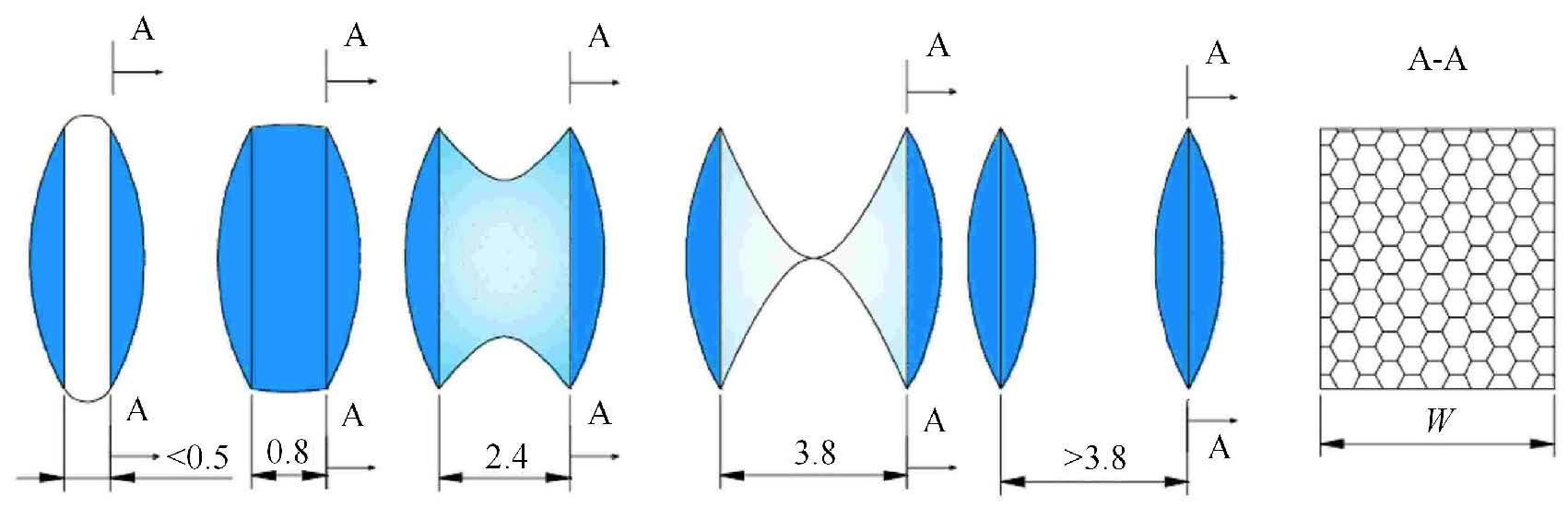
|
图5 纳米气泡桥形成/消失过程的示意图 Figure.5 Diagram of formation/disappearance of nanobubble bridge |
在伞状抽样后,得到如图6所示的平均力势曲线. 从中可以看出疏水力随两片石墨烯的间距增大而减小;并且在0.37∼0.8nm之间的梯度明显大于0.8∼3.8nm之间的梯度.这表明0∼0.8nm间的疏水力由低密度的纳米气泡桥引起,而0.8∼3.8nm间的疏水力由饱和纳米气泡桥导致.当两片石墨烯的间距大于约3.8nm时,平均力势曲线的梯度变为0,说明气泡桥断裂疏水力消失.
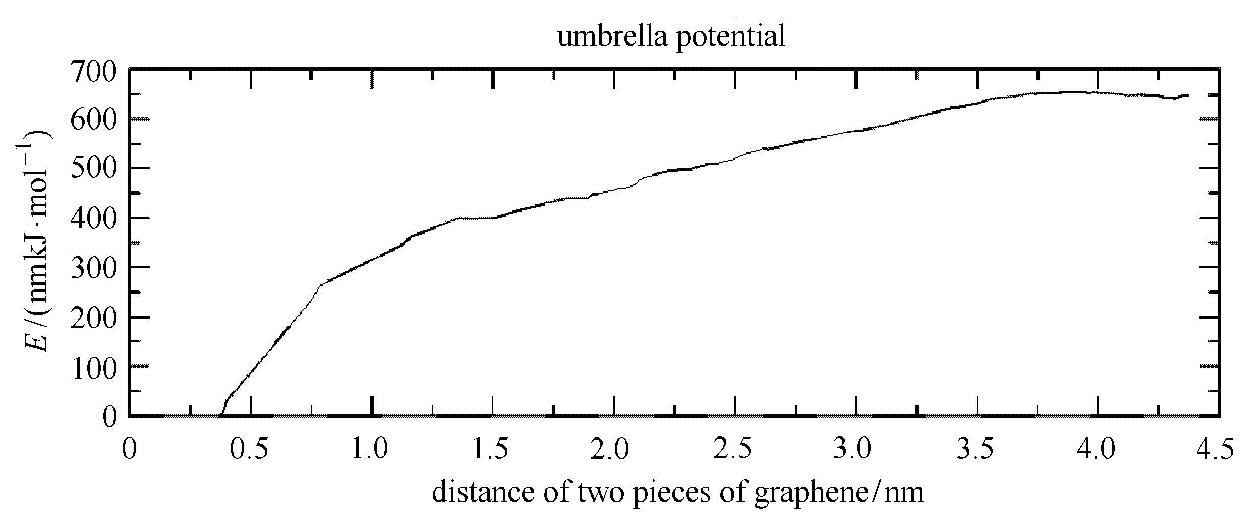
|
图6 有气体溶解的情况,两片石墨烯在水中相互作用的平均力势曲线 Figure.6 The calculated PMF curve for two pieces of graphene in the expansion with gas phase |
两片石墨烯在有气体溶解的水中和无气体溶解的水中的疏水引力都是由纳米气泡桥引起的,其中:
1)当石墨烯间距小于0.5nm时,无论水中是否溶解有气体疏水引力都是由真空纳米气泡桥引起的,这个结论与相关研究结果基本一致[31].
(2)当石墨烯间距大于0.5nm时,没有气体溶解的水中,疏水引力是由水蒸气纳米气泡桥引起的;而在有气体溶解的水中疏水引力是由所溶气体纳米气泡桥引起的.
| [1] | 骆庆群, 杨洁明. 基于纳米气泡的煤炭浮选模型研究. 太原理工大学学报, 2014, 45(2): 201-209 (Luo Qingqun, Yang Jieming. Nanobubble-based coal flotation model. Journal of Taiyuan University of Technology, 2014, 45(2): 201-209 (in Chiniese)) |
| [2] | Luo QQ,Yang JM. Gas adsorption and accumulation on hydrophobic surfaces: Molecular dynamics simulations. Chinese Physics B,2015, 24(9): 392-398 |
| [3] | Alsawafta M, Badilescu S, Truong VV, et al. The effect of hydrogen nanobubbles on the morphology of gold-gelatin bionanocomposite films and their optical properties. Nanotechnology, 2012, 23(6):65305-65313 |
| [4] | Bunkin NF, Yurchenko SO, Suyazov NV, et al. Structure of the nanobubble clusters of dissolved air in liquid media. Journal of Biological Physics, 2012, 38(1): 121-152 |
| [5] | Hampton MA, Nguyen AV. Nanobubbles and the nanobubble bridging capillary force. Advances in Colloid and Interface Science,2010, 154(1-2): 30-55 |
| [6] | Rtsadl J. Nanobubbles and micropancakes: gaseous domains on immersed substrates. Journal of Physics: Condensed Matter, 2011,23(13): 1296-1323 |
| [7] | Seddon J R T, Lohse D. Nanobubbles and micropancakes: gaseous domains on immersed substrates. Journal of Physics-Condensed Matter, 2011, 23(13): 1296-1323 |
| [8] | White ER, Mecklenburg M, Singer SB, et al. Imaging nanobubbles in water with scanning transmission electron microscopy. Applied Physics Express, 2011, 4(55201): 55201-55203 |
| [9] | Attard P. Thermodynamic analysis of bridging bubbles and a quantitative comparison with the measured hydrophobic attraction. Langmuir,2000, 16(10): 4455-4466 |
| [10] | Attard P. Nanobubbles and the hydrophobic attraction. Advances in Colloid and Interface Science, 2003, 104(104: 75-91 |
| [11] | Hampton MA, Donose BC, Nguyen AV. Effect of alcohol-water exchange and surface scanning on nanobubbles and the attraction between hydrophobic surfaces. Journal of Colloid and Interface Science,2008, 325(1): 267-274 |
| [12] | Ishida N, Sakamoto M, Miyahara M, et al. Attraction between hydrophobic surfaces with and without gas phase. Langmuir, 2000,16(13): 5681-5687 |
| [13] | Yakubov GE, Butt HJ, Vinogradova OI. Interaction forces between hydrophobic surfaces. attractive jump as an indication of formation of "stable" submicrocavities. The Journal of Physical Chemistry B,2000, 104(15): 3407-3410 |
| [14] | Mahnke J, Stearnes JA, Hayes R, et al. The influence of dissolved gas on the interactions between surfaces of different hydrophobicity in aqueous media Part I. Measurement of interaction forces. Physical Chemistry Chemical Physics, 1999, 1(11): 2793-2798 |
| [15] | Thormann E, Simonsen A C, Hansen P L, et al. Interactions between a polystyrene particle and hydrophilic and hydrophobic surfaces in aqueous solutions. Langmuir, 2008, 24(14): 7278-7284 |
| [16] | Koishi T, Yoo S, Yasuoka K, et al. Nanoscale hydrophobic interaction and nanobubble nucleation. Physical Review Letters, 2004,93(18): 10445-10458 |
| [17] | Duan Y, Wu C, Chowdhury S, et al. A point-charge force field for molecular mechanics simulations of proteins based on condensedphase quantum mechanical calculations. Journal of Computational Chemistry, 2003, 24(16): 1999-2012 |
| [18] | Cornell WD, Cieplak P, Bayly CI, et al. A second generation force field for the simulation of proteins, nucleic acids, and organic molecules. Journal of the American Chemical Society, 1995,117(19): 5179-5197 |
| [19] | Berendsen HJC, Postma JPM, Van Gunsteren WF, et al. Molecular dynamics with coupling to an external bath. The Journal of Chemical Physics, 1984, 81(8): 3684-3690 |
| [20] | Nosé S. A unified formulation of the constant temperature molecular dynamics methods. The Journal of Chemical Physics, 1984, 81(1):511-519 |
| [21] | Hoover W G. Canonical dynamics: equilibrium phase-space distributions. Physical Review A, 1985, 31(3): 1695-1697 |
| [22] | Nosé S, Klein ML. Constant pressure molecular dynamics for molecular systems. Molecular Physics, 1983, 50(5): 1055-1076 |
| [23] | Parrinello M, Rahman A. Polymorphic transitions in single crystals: A new molecular dynamics method. Journal of Applied Physics,1981, 52(12): 7182-7190 |
| [24] | Patey GN, Valleau JP. The free energy of spheres with dipoles: Monte Carlo with multistage sampling. Chemical Physics Letters,1973, 21(2): 297-300> |
| [25] | Torrie GM, Valleau JP. Nonphysical sampling distributions in Monte Carlo free-energy estimation: Umbrella sampling. Journal of Computational Physics, 1977, 23(2): 187-199 |
| [26] | Hub JS, De Groot BL, Van Der Spoel D. A free weighted histogram analysis implementation including robust error and autocorrelation estimates. Journal of Chemical Theory and Computation, 2010,6(12): 3713-3720 |
| [27] | Hummer G, Garde S. An information theory model of hydrophobic interactions. Proceedings of the National Academy of Sciences of the United States of America, 1996, 93(17): 8951-8955 |
| [28] | Israelachvili JN. Intermolecular and surface forces. Academic Press,1991, 42-44(3): 59-65 |
| [29] | 赵亚溥. 表面与界面物理力学. 北京: 科学出版社, 2012 |
| [30] | Zhao Yapu. Physical Mechanics of Surfaces and Interfaces. Beijing: Science Press, 2012 (in Chinese) |
| [31] | Yuan QZ, Zhao YP. Precursor film in dynamic wetting, electrowetting, and electro-elasto-capillarity. Physical Review Letters, 2010,104(24): 246101-246107 |
 2016, Vol. 48
2016, Vol. 48